How the Filta-Max system works
The Filta-Max module comprises multiple layers of open-cell foam discs that are compressed to create a filter with a nominal pore size to effectively trap Cryptosporidium and Giardia. After sampling, the filter is processed by releasing the compression of the foam discs and washing, enabling all captured material to be recovered. It is a simple yet extremely efficient system.
Filtration
To sample, the filter module is placed in a reusable housing, which is attached to the required sampling source. A head pressure of 0.5 Bar (7.5 psi) is required to create a flow, and the recommended operating pressure is 5 Bar (75 psi). This pressure will create a flow rate of 4 liters per minute, which is particularly beneficial when sampling large volumes of water.

Recovery
After filtration, the module is removed from the housing and placed in the wash station. IDEXX provides both an automatic wash station and a manual version. The foam discs are decompressed by removing a bolt in the module and then washed with an elution buffer using expansion/compression cycles.
The volume of buffer is then reduced to between 25 and 50 mL by concentrating through a membrane, negating the need for high-volume centrifugation.


 Connect the housing to the required water source for sampling
Connect the housing to the required water source for sampling Remove the filter module from the housing.
Remove the filter module from the housing.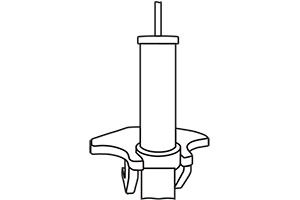 Place the module in a wash station and wash with elution buffer.
Place the module in a wash station and wash with elution buffer.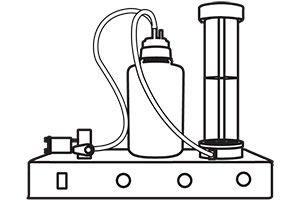 Concentrate the wash buffer using a 3-µm membrane.
Concentrate the wash buffer using a 3-µm membrane.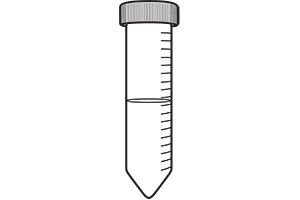 Repeat steps 3 and 4 to give a final volume of 25 mL.
Repeat steps 3 and 4 to give a final volume of 25 mL.